Difference between Acid and Base
Key difference: Acids and bases are two types of corrosive substances. Any substance with a pH value between 0 up to 7 is considered acidic, whereas a pH value of 7 to 14 is a base.
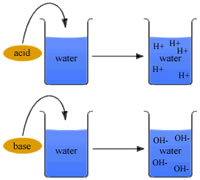
Acids are ionic compounds that break apart in water to form a hydrogen ion (H+). Ionic compounds are a compound with a positive or negative charge. Bases, on the other hand are ionic compounds that break apart to form a negatively charged hydroxide ion (OH-) in water.
 Acids
and bases are two types of corrosive substances. A corrosive substance
is a substance that will destroy and damage other substances with which
it comes into contact. Corrosive substances are known to have strong
reactions with variety of materials, including metals and various
organic compounds.
Acids
and bases are two types of corrosive substances. A corrosive substance
is a substance that will destroy and damage other substances with which
it comes into contact. Corrosive substances are known to have strong
reactions with variety of materials, including metals and various
organic compounds.All chemical compounds have a pH value. The pH scale ranges from 0 to 14. It is the measure of concentration of hydrogen ions in a solution. Any substance above the neutral pH is considered a base, whereas any substance below the neutral pH is considered an acid. The neutral pH is the pH of pure water, i.e. 7. So, any substance with a pH value between 0 up to 7 is considered acidic, whereas a pH value of 7 to 14 is a base.
Acids are ionic compounds that break apart in water to form a hydrogen ion (H+). Ionic compounds are a compound with a positive or negative charge. Bases, on the other hand are ionic compounds that break apart to form a negatively charged hydroxide ion (OH-) in water. The strength of an acid or base is determined on the basis of how many of their respective ions do they release; the more ions, the stronger they are.
Furthermore, if acids and bases of same strengths are combined they tend to produce a salt and water. For example: HCl (acid) + NaOh (base) = NaCl (household salt) + H2O (water)
Characteristics of Acids:
- taste sour when they are eaten
- can sting the skin when they are touched
- can corrode (or eat away at) metals and skin
- can be used as a reactant during electrolysis due to the presence of mobile ions
- turn blue litmus paper red
- are studied in chemistry and biology
- turn red or orange on universal indicator
Acids can be classified into:
- Strong acids — the most common are sulfuric acid, nitric acid and hydrochloric acid (H2SO4, HNO3 and HCl, respectively).
- Some concentrated weak acids, for example formic acid and acetic acid
- Strong Lewis acids such as anhydrous aluminum chloride and boron trifluoride
- Lewis acids with specific reactivity, e.g. solutions of zinc chloride
- Extremely strong acids (superacids)
 Characteristics of Bases (Alkalis):
Characteristics of Bases (Alkalis):- Bitter taste (opposed to sour taste of acids)
- Slimy, or soapy feel on fingers
- Many bases react with acids and precipitate salts.
- Strong bases may react violently with acids. An acid spill can be safely neutralised by using a mild base.
- Bases turn red litmus paper blue
- Bases are substances that contain metal oxides or hydroxides
- Bases which are soluble in water form alkalis (soluble bases)
Bases can be classified into:
- Caustics or alkalis, such as sodium hydroxide (NaOH) and potassium hydroxide (KOH)
- Alkali metals in the metallic form (e.g. elemental sodium), and hydrides of alkali and alkaline earth metals, such as sodium hydride, function as strong bases and hydrate to give caustics
- Extremely strong bases (superbases) such as alkoxides, metal amides (e.g. sodium amide) and organometallic bases such as butyllithium
- Some concentrated weak bases, such as ammonia when anhydrous or in a concentrated solution
Acid
|
Base
|
|
| Arrhenius Definition | An acid is any chemical compound which when dissolved in water gives a solution with a hydrogen ion activity greater than in pure water. | A base is an aqueous substance that can accept hydrogen ions. |
| Bronstead Lowry Definition | An acid is an substance which donates a proton. | A base is any substance which accepts a proton. |
| pH value | Less than 7.0 | Greater than 7.0 |
| Litmus paper | Blue litmus paper turns red | red litmus paper turns blue |
| Phenolphthalein | Remains colorless | Makes the solution pink |
| Dissociation (in water) | Acids free hydrogen ions (H+) when mixed with water. | Bases free hydroxide ions (OH-) when mixed with water. |
| Chemical Formula | An acid has a chemical formula with H at the beginning of it. For example, HCl (Hydrochloric Acid). There is one exception to his rule, CH3COOH = Acetic Acid (vinegar). | A base has a chemical formula with OH at the end of it. For example, NaOH (Sodium Hydroxide). |


ليست هناك تعليقات:
إرسال تعليق